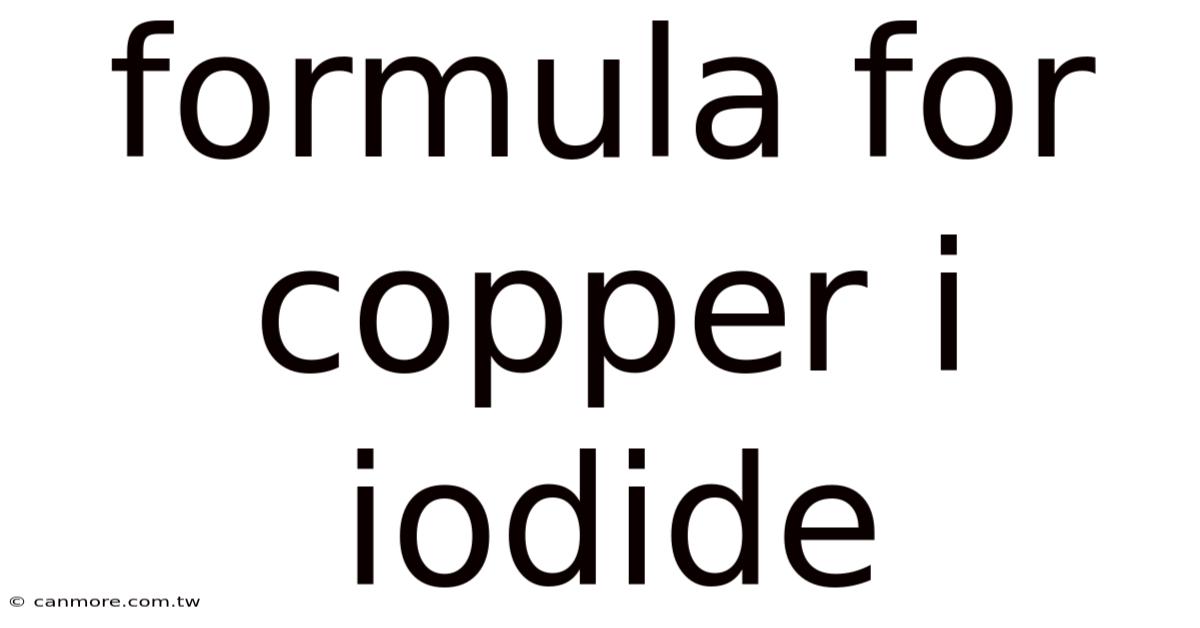The Formula for Copper(I) Iodide: Synthesis, Properties, and Applications
Copper(I) iodide, a fascinating inorganic compound, finds applications in diverse fields, from medicine to material science. Understanding its chemical formula, synthesis methods, properties, and applications is crucial for appreciating its significance. This full breakdown walks through the intricacies of copper(I) iodide, providing a detailed exploration suitable for students, researchers, and anyone curious about this remarkable compound The details matter here..
Understanding the Chemical Formula: CuI
The chemical formula for copper(I) iodide is simply CuI. This notation signifies that the compound consists of one copper(I) ion (Cu⁺) and one iodide ion (I⁻). The Roman numeral I in parentheses indicates the oxidation state of copper, which is +1 in this compound. It's essential to distinguish this from copper(II) iodide, which would have a different formula and properties. Understanding oxidation states is crucial in predicting the chemical behavior of this and other transition metal compounds But it adds up..
Synthesis of Copper(I) Iodide: Diverse Approaches
Several methods exist for synthesizing copper(I) iodide, each with its advantages and disadvantages. The choice of method often depends on the desired purity, scale of production, and available resources.
Method 1: Direct Reaction of Copper(II) Sulfate and Potassium Iodide
One common approach involves the reaction between copper(II) sulfate (CuSO₄) and potassium iodide (KI). This reaction is an example of a redox reaction, where copper(II) is reduced to copper(I) and iodide is oxidized to iodine.
The balanced chemical equation is:
2CuSO₄ + 4KI → 2CuI + I₂ + 2K₂SO₄
This reaction produces a precipitate of copper(I) iodide, along with iodine (I₂) and potassium sulfate (K₂SO₄). The iodine byproduct needs to be removed through filtration and washing. The precipitated CuI is then typically dried to obtain the final product. This method is relatively straightforward but generates iodine waste that needs careful handling.
Method 2: Reaction of Copper(II) Chloride and Potassium Iodide
A similar redox reaction can be achieved using copper(II) chloride (CuCl₂) instead of copper(II) sulfate. The balanced chemical equation is analogous:
2CuCl₂ + 4KI → 2CuI + I₂ + 4KCl
This method also produces iodine as a byproduct and requires subsequent purification steps. The choice between using copper(II) sulfate or chloride depends on availability and cost factors.
Method 3: Reaction of Copper(II) Salts with Sodium Thiosulfate and Potassium Iodide
A more controlled method involves the use of sodium thiosulfate (Na₂S₂O₃) as a reducing agent. The thiosulfate reacts with the iodine produced in the initial reaction, preventing its interference with the copper(I) iodide formation. This approach yields a purer product compared to direct reactions.
- 2Cu²⁺ + 4I⁻ → 2CuI + I₂
- 2Na₂S₂O₃ + I₂ → 2NaI + Na₂S₄O₆
This two-step process offers better control over the reaction, leading to a higher yield and purity of copper(I) iodide. The sodium tetrathionate (Na₂S₄O₆) byproduct is soluble and easily separated from the CuI precipitate The details matter here..
Method 4: Controlled Precipitation from Copper(I) Solution
A more specialized method involves the preparation of a copper(I) solution, typically by reducing a copper(II) solution with a suitable reducing agent such as ascorbic acid or hydrazine. On the flip side, this copper(I) solution is then treated with a potassium iodide solution to precipitate CuI. This approach allows for finer control over the particle size and morphology of the resulting copper(I) iodide That's the whole idea..
The choice of synthesis method depends on various factors such as desired purity, scale of production, and cost-effectiveness. Each method offers a unique balance between simplicity and product quality And that's really what it comes down to..
Properties of Copper(I) Iodide: A Closer Look
Copper(I) iodide exhibits several important physical and chemical properties:
- Appearance: It's typically a white to pale yellow powder. That said, exposure to light can cause discoloration due to the formation of iodine.
- Solubility: CuI is practically insoluble in water, making it easy to isolate through precipitation. It has limited solubility in various organic solvents.
- Melting Point: CuI melts at a relatively high temperature (around 606°C).
- Crystal Structure: It adopts a zinc blende structure, a common crystal structure in many binary compounds. This structure dictates many of its physical properties.
- Reactivity: While relatively stable, copper(I) iodide can react with strong oxidizing agents, leading to oxidation of copper(I) to copper(II). It is also sensitive to light, as mentioned earlier.
Applications of Copper(I) Iodide: A Wide Spectrum
The unique properties of copper(I) iodide have led to its widespread use in several applications:
- Cloud Seeding: CuI is used as a cloud seeding agent, enhancing precipitation in areas experiencing drought. Tiny CuI particles act as ice nuclei, facilitating ice crystal formation in supercooled clouds.
- Medicine: It finds limited use in certain medical applications. Even so, research is ongoing to explore its potential in various therapeutic contexts.
- Catalysis: CuI serves as a catalyst in several organic reactions, including coupling reactions and other synthetic transformations. Its ability to allow electron transfer makes it valuable in various catalytic processes.
- Photography: Historically, CuI has been used in photography, although its application has diminished with the advent of digital photography.
- Material Science: It is employed in the synthesis of various materials, including semiconductor materials and other functional materials. Its role in the design and fabrication of advanced materials is constantly expanding.
- Antimicrobial agent: Recent research also points to the potential of CuI as an antimicrobial agent due to its ability to disrupt the metabolic processes of microorganisms.
- Solar cells: Copper(I) iodide shows promise as a component in the development of efficient and cost-effective solar cells. Its ability to help with charge transport makes it a promising candidate for this application.
Frequently Asked Questions (FAQ)
Q1: Is Copper(I) Iodide toxic?
A1: Like many inorganic compounds, CuI can be toxic if ingested or inhaled in large quantities. Appropriate safety measures should be taken during handling and disposal.
Q2: How is the purity of synthesized CuI assessed?
A2: Purity can be determined through various analytical techniques, including X-ray diffraction (XRD) to confirm the crystal structure, elemental analysis to determine the copper and iodine content, and other methods to check for impurities It's one of those things that adds up..
Q3: Why is CuI sensitive to light?
A3: The sensitivity to light stems from the ease with which copper(I) can be oxidized to copper(II) in the presence of light, accompanied by the release of iodine. This leads to discoloration and potential changes in its chemical properties Simple, but easy to overlook. Turns out it matters..
Q4: What are the environmental concerns related to the use of CuI?
A4: Although generally considered to have relatively low environmental impact compared to some other compounds, appropriate disposal methods are necessary to prevent potential contamination of water sources or soil.
Q5: What is the difference between CuI and CuI₂?
A5: CuI refers to copper(I) iodide, where copper has an oxidation state of +1. CuI₂ (copper(II) iodide) would represent a compound where copper has an oxidation state of +2, but this compound is unstable and readily decomposes into CuI and I₂.
Conclusion: A Versatile Compound with Expanding Applications
Copper(I) iodide, with its simple chemical formula CuI, exhibits a surprisingly diverse range of properties and applications. In real terms, from its role in cloud seeding to its potential in catalysis and material science, CuI continues to be an area of active research and development. Here's the thing — understanding its synthesis, properties, and applications is crucial for appreciating its significance in various scientific and technological domains. Further research into its potential in medicine, solar energy, and other emerging fields promises even wider applications in the future. Its unique reactivity and relatively simple synthesis methods make it a valuable compound in both academic and industrial settings, highlighting its ongoing importance across several scientific disciplines.